| Principle |
Chromatographic Immunoassay |
| Format |
Cassette |
| Specimen |
WB |
| Certificate |
CE |
| Reading Time |
10 minutes |
| Pack |
10 T |
| Storage Temperature |
2-30°C |
| Shelf Life |
2 Years |
| Sensitivity |
94.10% |
| Specificity |
92.00% |
| Accuracy |
93.80% |
| Cut-Off |
10-100 ng/ml |
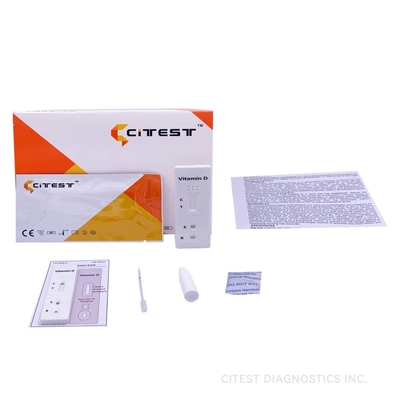
Vitamin D Rapid Test Cassette ,Fingerstick Whole blood,detection of 25-hydroxyvitamin D (25 (OH) D)
Vitamin D (VD for short) is a fat-soluble vitamin, a cyclopentane polyhydrophenanthrene compound, a group of vitamins structurally related to sterols and functionally preventing rickets, most notably vitamin D3 and D2. The former is made from 7-dehydrocholesterol in the human subcutis by UV irradiation. The latter is made from ergocalciferol contained in plants or yeast by ultraviolet irradiation.
The main function of vitamin D is to promote the absorption of calcium and phosphorus by the mucosal cells of the small intestine. Calcium ion absorption in the intestine requires a calcium-binding protein, and 1,25-dihydroxyvitamin D3 can induce the synthesis of this protein and promote Ca2+ absorption, which can also promote the renewal of calcium salts and new bone production, and also promote phosphorus absorption and renal tubular cell reabsorption of calcium and phosphorus, so it can increase the blood calcium and phosphorus concentration, which is beneficial to new bone production and calcification.
In addition, vitamin D also promotes skin cell growth, differentiation and regulation of immune function. In general, adults with regular exposure to sunlight do not suffer from deficiency, while infants, pregnant women, lactating mothers and elderly people who do not often go outdoors should increase their supply of vitamin D to 10 μg (equivalent to 400 international units) per day. Vitamin D deficiency can lead to rickets in children and osteomalacia in adults.
Food sources are relatively high in fat-containing sea fish, animal liver, egg yolk, cream, and high in cod liver oil

A rapid test for the semi-quantitative detection of 25-hydroxyvitamin D in human fingerstick Whole Blood.
For professional in vitro diagnostic use only.
INTENDED USE
The Vitamin D Rapid Test Cassette is a rapid chromatographic immunoassay for the semi-quantitative detection of 25-hydroxyvitamin D (25 (OH) D) in human fingerstick Whole blood at a cut-off concentration of 30 ± 4ng/mL. This assay provides a preliminary diagnostic test result and can be used to screening for Vitamin D deficiency.
SUMMARY
Vitamin D refers to a group of fat-soluble secosteroids responsible for increasing intestinal absorption of calcium, iron, magnesium, phosphate and zinc. In humans, the most important compounds in this group are vitamin D3 and vitamin D2.[1] Vitamin D3 is naturally produced in the human skin through the exposure to ultraviolet light and Vitamin D2 is mainly obtained from foods. Vitamin D is transported to the liver where it is metabolized to 25-hydroxy Vitamin D. In medicine, a 25-hydroxy Vitamin D blood test is used to determine Vitamin D concentration in the body.
The blood concentration of 25-hydroxy Vitamin D (including D2 and D3) is considered the best indicator of Vitamin D status. Vitamin D deficiency is now recognized as a global epidemic.[2] Virtually every cell in our body has Receptors for Vitamin D, meaning that they all require “Sufficient” Level of Vitamin D for adequate functioning.
The health risks associated with Vitamin deficiency are far more severe than previously thought. Vitamin deficiency has been linked to various serious diseases: Osteoporosis, Osteomalacia, Multiple Sclerosis, Cardiovascular Diseases, Pregnancy Complications, Diabetes, Depression, Strokes, Autoimmune Diseases, Flu, Different Cancers, Infectious Diseases, Alzheimer, Obesity and Higher Mortality etc.[3] Therefore, now detecting (25-OH) Vitamin D level is considered as “Medically Necessary Screening Test”, and maintaining sufficient levels not just to improve bone health, but to improve overall health and well-being.
PRINCIPLE
The Vitamin D test is an immunoassay based on the principle of competitive binding. During testing, the mixture migrates upward on the membrane chromatographically by capillary action. The membrane is pre-coated with 25 (OH) D antigens on the test line region of the strip. During testing, 25 (OH) D present in the specimen will compete with 25 (OH) D on the test line for limited amount of anti-25 OH Vitamin D antibodies in the conjugate. The higher concentration of 25 (OH) D in the specimen, the lighter would be the T line. The result will be read according to Color card provided with the kit.
To serve as a procedural control, a colored line will always appear in the control line region indicating that proper volume of specimen has been added and membrane wicking has occurred.
REAGENTS
The test contains anti-25 OH Vitamin D antibody coated particles and 25-OH Vitamin D antigen coated on the membrane.
PRECAUTIONS
Please read all the information in this package insert before performing the test.
1. For professional in vitro diagnostic use only. Do not use after the expiration date.
2. The test should remain in the sealed pouch until ready to use.
3. All specimens should be considered potentially hazardous and handled in the same manner as an infectious agent.
4. The used test should be discarded according to local regulations.
DIRECTIONS FOR USE
Allow the test, specimen, buffer and/or controls to reach room temperature (15-30°C) prior to testing
1. Remove the test cassette from the sealed pouch and use it as soon as possible.
2. Place the cassette on a clean and level surface.
To use a Capillary droppers: Fill the capillary tube and transfer approximately 20uL of fingerstick whole blood specimen to the specimen area of test cassette, then add
2 drops of buffer and start the timer. See illustration below.
3. Wait for the colored line(s) to appear. Read results at 10 minutes by comparing the T line intensity with provided color card. Do not interpret the result after 20 minutes.
Note: It is suggested not to use the buffer, beyond 6 months after opening the vial.

INTERPRETATION OF RESULTS
(Please refer to the illustration and compare the T line intensity with “Vitamin D Color card” provided with the kit.)
Deficient: Two distinct colored lines appear. One is in the control region (C) and another should be in the test region (T). The line intensity in the test region (T) is equal to or darker than 10 ng/mL line depicted on color card provided with the kit.
Insufficient: Two colored lines appear. One is in the control region (C) and another should be in the test region (T). The line intensity in the test region (T) is darker than the 30 ng/mL line depicted on the color card provided with the kit and lighter than 10 ng/mL line depicted on Color card provided with the kit.
Sufficient: Two colored lines appear, one line should be always in the control region (C) and faint colored line appears in the test region (T). The line intensity in region (T) is darker than the 100 ng/mL line depicted on the Color card and lighter than 30 ng/mL line depicted on color card.
Note: Always compare the T line intensity with “Vitamin D Color card” and interpret results accordingly.
INVALID: Control line fails to appear. Insufficient specimen volume or incorrect procedural techniques are the most likely reasons for control line failure. Review the procedure and repeat the test with a new test. If the problem persists, discontinue using the test kit immediately and contact your local distributor.
QUALITY CONTROL
An internal procedural control is included in the test. A colored line appearing in the control line region (C) is an internal positive procedural control. It confirms sufficient specimen volume, adequate membrane wicking and correct procedural technique.
Control standards are not supplied with this kit; however, it is recommended that standard controls be tested as a good laboratory practice to confirm the test procedure and to verify proper test performance.

![]()
![]()